Abstract
The soybean technology MON 87701 × MON 89788, expressing Cry1Ac and conferring tolerance to glyphosate, has been widely adopted in Brazil since 2013. However, pest shifts or resistance evolution could reduce the benefits of this technology. To assess Cry1Ac soybean performance and understand the composition of lepidopteran pest species attacking soybeans, we implemented large-scale sampling of larvae on commercial soybean fields during the 2019 and 2020 crop seasons to compare with data collected prior to the introduction of Cry1Ac soybeans. Chrysodeixis includens was the main lepidopteran pest in non-Bt fields. More than 98% of larvae found in Cry1Ac soybean were Spodoptera spp., although the numbers of Spodoptera were similar between Cry1Ac soybean and non-Bt fields. Cry1Ac soybean provided a high level of protection against Anticarsia gemmatalis, C. includens, Chloridea virescens and Helicoverpa spp. Significant reductions in insecticide sprays for lepidopteran control in soybean were observed from 2012 to 2019. Our study showed that C. includens and A. gemmatalis continue to be primary lepidopteran pests of soybean in Brazil and that Cry1Ac soybean continues to effectively manage the target lepidopteran pests. However, there was an increase in the relative abundance of non-target Spodoptera spp. larvae in both non-Bt and Cry1Ac soybeans.
Similar content being viewed by others
Introduction
Brazil is a top producer of soybean (Glycine max (L.) Merrill)1, and a turning point in the commercial cultivation of soybean in Brazil was the expansion of soybean production from the South (subtropical climate) into areas in the savannahs (i.e., Cerrado) in the Central-West region of the country (tropical climate)2. This expansion was largely enabled by technological advances ranging from better soil management and fertilization practices to the development of soybean varieties adapted to equatorial latitudes3. Moreover, the expansion of no-till systems, the use of better planting and harvesting equipment, and the adoption of transgenic soybeans to assist in weed control made important contributions to increases in the national average soybean yield in Brazil1,3,4,5. As soybean production in Brazil transformed into a highly structured and organized large-scale business operation primarily targeting export markets, the need to adopt simple, low-cost agronomic practices for controlling insects caused an increase in the number of insecticide sprays required6,7,8.
In this context, the soybean technology MON 87701 × MON 89788 (Intacta RR2 PRO®), expressing the Cry1Ac insecticidal protein (event MON 87701) and conferring tolerance to glyphosate (event MON 89788), was commercially launched and became available to farmers in Brazil in 2013. The adoption and use of Cry1Ac soybean by Brazilian farmers increased from 1.2 million hectares in the 2013/14 cropping season to 21.9 million hectares in the 2017/18 cropping season9. The acceptance of this soybean technology by Brazilian farmers can be attributed to the cost-effective and simpler weed control enabled by the tolerance to glyphosate, coupled with higher yields from a combination of better pest and weed management10. Cry1Ac soybean provides high-level protection against the major soybean lepidopteran pests, including Anticarsia gemmatalis (Hübner, 1818) (Lepidoptera: Erebidae), Chrysodeixis includens (Walker [1858]) (Lepidoptera: Noctuidae), Chloridea virescens (Fabricius, 1777) (Lepidoptera: Noctuidae) and Helicoverpa armigera (Hübner, 1808) (Lepidoptera: Noctuidae)11,12,13,14. Despite its benefits to soybean pest management, Cry1Ac soybean does not confer protection against the main species of Spodoptera found in Brazil: Spodoptera frugiperda (J.E. Smith, 1797) (Lepidoptera: Noctuidae), Spodoptera eridania (Stoll, 1782) (Lepidoptera: Noctuidae) and Spodoptera cosmioides (Walker, 1898) (Lepidoptera: Noctuidae)15.
Beyond the direct benefit of controlling target pests, Bt crops such as Cry1Ac soybean have the potential to provide additional benefits to insect management in agricultural systems, including reduction in insecticide use10, compatibility with biocontrol measures16,17, and regional suppression of insect pest populations18,19,20,21. In particular, suppression of target pests after a long period of use of highly efficacious Bt technologies has been documented in Pectinophora gossypiella (Saund., 1844) (Lepidoptera: Gelechiidae), Ostrinia nubilalis (Hübner, 1796) (Lepidoptera: Pyralidae) and Helicoverpa zea (Boddie, 1850) (Lepidoptera: Noctuidae) in the USA18,19,21 and H. armigera in China20. Similarly, high efficacy of Cry1Ac soybean against lepidopteran pests such as C. includens and A. gemmatalis, the main soybean pests in Brazil12, resulted in fewer insecticide sprays to manage lepidopteran larvae after five years of commercial use in Brazil10. However, where a Bt technology is ineffective against non-target secondary pest species and or broad-spectrum insecticide use has decreased due to highly effective control of the target species, secondary pests may increase in abundance over time22,23.
In addition, resistance evolution by target pest populations can reduce the benefits of Bt crops24. The high-dose expression and refuge strategy was proposed to manage resistance of target pest populations to Cry1Ac soybean in Brazil12. Nevertheless, poor compliance with refuge recommendations has been a common factor in most cases of documented field-evolved resistance to Bt crops25,26,27,28,29. Moreover, the intensive use of agricultural land creates an environment conducive to the buildup of relatively large insect pest populations and multiple generations of pests potentially under selection of Bt crops or insecticides30,31. “Tropical agriculture” such as that practiced in Brazil is typically based on two or more crop seasons per year, allowing pest populations to go through multiple generations per year on Bt crops and consequently increasing selection pressure30. Therefore, understanding the performance of a Bt crop against target pests at the field level and determining whether non-target pests are increasing in abundance can inform the need for the adoption of appropriate Integrated Pest Management (IPM) practices in Brazil.
In this study, we carried out a two-year large-scale assessment on commercial soybean fields in Brazil after eight years of Cry1Ac soybean use with the goals of (a) evaluating Cry1Ac soybean performance and impacts on soybean pest management and (b) assessing the relative abundance of lepidopteran pest species attacking soybean fields and comparing these results to data collected prior to the commercial introduction of Cry1Ac soybeans.
Methods
Insect sampling and data collection
All insect collections were in accordance with the approval granted by the System of Authorization and Information on Biodiversity (SISBIO) of the Ministry of Environment to a contracted company responsible for the field sampling (PROMIP, Permit for scientific purpose activity: 61826, 61824).
Sampling prior to commercialization of Cry1Ac soybean
From 2011 (2010/11) to 2014 (2013/14), field sampling of lepidopteran larvae was carried out in plots of non-Bt (Roundup Ready—RR) soybean. Samples consisted of 10 beat sheets (length = 1 m) per location followed by identification of larvae. A total of 829 samples were taken across the states of Bahia (BA), Distrito Federal (DF), Goiás (GO), Mato Grosso (MT), Mato Grosso do Sul (MS), Minas Gerais (MG), Paraná (PR), Rio Grande do Sul (RS), Santa Catarina (SC), São Paulo (SP) and Tocantins (TO). These data from 2011–2014 provided a baseline for descriptive comparisons with the subsequent sampling described below but were not analyzed statistically.
Sampling after commercialization of Cry1Ac soybean
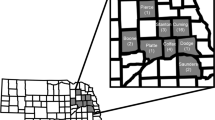
Lepidopteran larvae were sampled from commercial 399 soybean fields during the 2019 (2018/19) and 387 fields in 2020 (2019/20) cropping seasons (Fig. 1). Each location had a non-Bt (Roundup Ready—RR) soybean field and a Cry1Ac soybean (MON 87701 × MON 89788, Intacta RR2 PRO®) field. Samplings were conducted at early reproductive stages (R1–R4) and late reproductive stages (R5–R7). Larvae were sampled with a beat sheet (length = 1 m) and the sampling unit consisted of 10 beats in a zig-zag pattern per soybean reproductive stages. Additionally, for each beat sheet sampling, the level of defoliation in soybean was evaluated. To avoid border effects, sampling was initiated at a minimum of 20 m from the edge of the soybean fields in the Southern region of Brazil, where farms are smaller (average size of farms less than 100 ha), and 100 m in the Central-West and Northeast regions, where larger farms are common (average size of farms greater than 150 ha). For each location, sampling was done first in the non-Bt field: if at least 1 larva per meter was obtained, then samples were also taken from a nearby Cry1Ac soybean field at a similar plant growth stage to have a pair of neighboring fields with comparable incidence of lepidopteran pests. All fields were checked for Cry1Ac expression using QuickStix kits (Envirologix) to confirm the presence of Bt protein in Cry1Ac soybean plants and absence of this protein in non-Bt soybean plants. Larvae were transferred to 50-mL labeled conical centrifuge tubes containing propylene glycol. All tubes were then sent to the laboratory and kept in a freezer (− 20 °C) until identification. All lepidopteran larvae were identified based on Herzog32, Sosa-Gómez et al.33 and Gilligan and Passoa34.
Lepidopteran species composition sampled from non-Bt fields prior and after commercialization of Cry1Ac soybean
A descriptive analysis was made with the total insects sampled prior (2011–2014) and after (2019–2020) commercialization of Cry1Ac soybean. The relative number of each species for 2011–14 and 2019–20 was represented as a percentage of total.
Comparison of pest abundance on Cry1Ac soybean and non-Bt soybean
Fields with Cry1Ac soybean were paired with neighboring non-Bt fields (see “Insect sampling and data collection”). To compare larval counts in Cry1Ac soybean fields with counts in non-Bt soybean fields, a generalized linear mixed model with Poisson link was fit to the data separately for each species and plant growth stage (combined across seasons 2019 and 2020). If \({Y}_{ij}\) is the count for the ith field type in the jth pair, then \(E\left(\mathrm{log}\left({Y}_{ij}\right)\right)={u}_{j}\) for non-Bt fields, and \(E\left(\mathrm{log}\left({Y}_{ij}\right)\right)={\tau }{+u}_{j}\) for Cry1Ac soybean fields, where \({u}_{j}\sim N\left(0,{\sigma }^{2}\right)\) is the effect of the jth pair and \(\mathrm{exp}(\tau )\) is the relative larvae count in Cry1Ac soybean fields. Analyses were performed with R statistical software—R version 4.0.235.
Pest species contributions to defoliation
To determine the relative contribution of individual species to defoliation, multiple regression was performed of defoliation against counts of A. gemmatalis, C. includens, C. virescens, Helicoverpa spp., Rachiplusia nu (Guenée) (Lepidoptera: Noctuidae), S. cosmioides, S. eridania and S. frugiperda. Regression was conducted separately for Cry1Ac soybean and non-Bt soybean, and for early and late growth reproductive stages, combined across the 2019 and 2020 seasons. The regression coefficient for a given species can be interpreted as the percentage increase in defoliation for each individual larva present. Thus, species with large coefficients contributed more to defoliation than did species with small coefficients. Analyses were performed with R statistical software—R version 4.0.235.
Geographic variation in soybean pest abundance
Sampling locations were grouped according to Embrapa’s soybean variety regionalization36. These groupings are called “edaphoclimatic regions” and “soybean macroregions” and are based on agroecological zones, Köppen climate classification for Brazil, technical recommendations for soybean production, soybean research meeting documents, and contributions of research institutes36. To characterize geographic variation in pest abundance, random effects for edaphoclimatic regions were estimated using a linear mixed-effects model for larval count data with Poisson link, with edaphoclimatic region nested within soybean macroregion. The abundance was estimated based on non-Bt soybean larval sampling. The edaphoclimatic region estimates were color-coded in choropleth maps. Analyses were performed with R statistical software—R version 4.0.235.
Insecticide use on soybean fields in Brazil
Data on use of insecticide sprays to manage lepidopteran larvae across mesoregions for the 2013 to 2019 cropping seasons were obtained from the AMIS Kleffmann Group database (2013–2018) and BIP Spark (2019). A linear regression analysis was performed with number of insecticide sprays as a function of cropping season. The number of insecticide sprays for lepidopteran control in every mesoregion of soybean planting area was log-transformed. Analyses were performed in GraphPad Prism 8 (GraphPad Software, San Diego, CA, USA).
Results
Lepidopteran species composition sampled from non-Bt fields
The percentage of each pest species relative to total larvae sampled in commercial (non-Bt) soybean fields prior to the commercialization of Cry1Ac soybean (2011 to 2014 cropping seasons; hereafter “pre-commercial period”) and total larvae sampled in commercial non-Bt soybean fields during the post-commercial period of Cry1Ac soybean (2019 and 2020 cropping seasons; hereafter “post-commercial period”) is shown in Fig. 2. Of the 16,277 lepidopteran larvae sampled in non-Bt fields during the pre-commercial period, more than 90% were C. includens and A. gemmatalis (Fig. 2A). The importance of these two species in non-Bt soybean fields was maintained during the post-commercial period, in which C. includens and A. gemmatalis represented more than 70% of 12,676 insects sampled (Fig. 2B). Small numbers of other species such Helicoverpa spp., R. nu and C. virescens were present, together representing less than 6% and less than 5% of the total samples during the pre- and post-commercial periods, respectively. Spodoptera species, represented mostly by S. frugiperda, S eridania and S. cosmioides, also were found in low numbers during the pre-commercial period. Representing only 3% of 12,676 insects sampled during the pre-commercial period, S. eridania was the predominant species sampled. However, Spodoptera species represented more than 23% of the total insects sampled in non-Bt soybean fields during the post-commercial period (Fig. 2B).
The absolute numbers of S. cosmioides, S. eridania and S. frugiperda found in Cry1Ac and non-Bt soybean were similar (Fig. 3A). Of the 1,376 and 1,122 total insects sampled in Cry1Ac soybean at the early and late reproductive stages, respectively, more than 98% were S. cosmioides, S. eridania and S. frugiperda (Fig. 3B). More S. frugiperda were observed at the early reproductive stage and more S. eridania at the late reproductive stage in both soybean types (Fig. 3B). For non-Bt soybean, 12,676 larvae were identified, of which 51.32% and 68.26% were C. includens at the early and late reproductive stages, respectively (Fig. 3C). S. frugiperda and A. gemmatalis represented similar percentages of the total insects from non-Bt soybean at the early reproductive stage (18.21% and 18.24%, respectively) and late reproductive stage (7.95% and 5.93%, respectively) (Fig. 3C).
Lepidopteran species composition of sampled larvae from Cry1Ac soybean and non-Bt fields in the 2019 and 2020 cropping seasons. (A) Total number of lepidopteran larvae sampled in Cry1Ac soybean and non-Bt soybean fields. (B) Larvae species composition from Cry1Ac soybean fields in early and late stage. (C) Larvae species composition from non-Bt soybean fields in early and late stage.
The median percentage defoliation in Cry1Ac soybean fields was lower than in non-Bt soybean fields in all but one comparison. For 2019, the Cry1Ac soybean median defoliation was 2.5% and 4.7% at the early and late reproductive stages, respectively, whereas median defoliation in non-Bt soybean fields was 8.6% and 13.7% at the early and late reproductive stages, respectively (Fig. 4). The 75th percentiles for defoliation in Cry1Ac soybean were 5% and 6.5% and for non-Bt soybean were 13.3% and 21.5% at the early and late reproductive stages, respectively (Fig. 4). In 2020, median defoliation in both Cry1Ac soybean and non-Bt soybean was 5% at the early reproductive stage, and 5% and 10%, respectively, at the late reproductive stage (Fig. 4). The 75th percentiles for defoliation at the late reproductive stage were 9.8% and 15% for Cry1Ac soybean and non-Bt soybean, respectively (Fig. 4).
Comparison of pest abundance on Cry1Ac soybean and non-Bt soybean
Larval abundance in Cry1Ac soybean fields relative to non-Bt soybean fields in the 2019 and 2020 seasons, and 95% confidence intervals, are presented in Table 1. Based on these analyses, Cry1Ac soybean provided high control (relative abundance < 0.02) of A. gemmatalis, C. includens, C. virescens and Helicoverpa spp.; minimal control of S. eridania and S. frugiperda; and no control of S. cosmioides (Table 1). R. nu larvae were rare in samples from both non-Bt and Cry1Ac soybean fields. The relative Cry1Ac soybean abundance values were similar among early and late reproductive stages within each species, with the exception that S. cosmioides was more prevalent at the early reproductive stage (Table 1).
Pest species contributions to defoliation
Table 2 gives regression coefficient estimates for early and late reproductive stage non-Bt and Cry1Ac soybean. As described earlier, these coefficients represent the percentage increase in defoliation caused by each larva of a species. In non-Bt soybean fields, A. gemmatalis, C. includens, Helicoverpa spp., S. cosmioides, S. eridania and S. frugiperda contributed to defoliation at both early and late reproductive stages (P < 0.05) (Table 2). The highest coefficients observed for non-Bt soybean were for S. cosmioides: 1.385 and 2.136 for early and late reproductive stages, respectively (Table 2). Comparing S. cosmioides with S. frugiperda on early-reproductive-stage soybean, for example, the estimated coefficients were 1.385 and 0.245, respectively, indicating that an individual S. cosmioides larva caused 1.385/0.245 = 5.65 times the damage caused by an individual S. frugiperda larva. By that same logic, S. cosmioides caused 1.7 to 6.7 times the damage per larva caused by A. gemmatalis, C. includens, Helicoverpa spp. and S. eridania at the early reproductive stage. At the late reproductive stage, S. cosmioides caused 1.9 to 4.3 times the damage per larva caused by A. gemmatalis, C. includens, Helicoverpa spp., S. eridania and S. frugiperda.
For Cry1Ac soybean fields, only S. cosmioides, S. eridania and S. frugiperda contributed to defoliation (P < 0.05) (Table 2). The other species were controlled by Cry1Ac soybean, as described in the previous section. S. cosmioides had the highest coefficients in Cry1Ac soybean: 0.623 and 2.121 for early and late reproductive stages, respectively. Each S. cosmioides larva caused 2.0 and 4.5 times the damage at the early reproductive stage and 4.9 and 2.7 times the damage at the late reproductive stage caused by individual S. eridania and S. frugiperda larvae, respectively.
Geographic variation in soybean pest abundance
Visualization of pest abundance in non-Bt soybean by species in Figs. 5 and 6 shows that C. includens was present at high levels in all of the soybean-producing regions sampled in Brazil. A. gemmatalis and S. eridania abundance varied among seasons and growth stages but both species were often present at high levels. S. frugiperda abundance was lower in 2019 than in 2020, when there was high infestation in northern regions (Figs. 5 and 6). Abundances of C. virescens, Helicoverpa spp. and S. cosmioides were lower across the regions evaluated when compared to C. includens, A. gemmatalis, S. eridania and S. frugiperda (Figs. 5 and 6).
Insecticide spray usage on soybeans fields in Brazil
The number of sprays for management of lepidopteran larvae over mesoregions decreased from an average of 3.5 in 2012 to 2.45 in 2019 cropping season (F = 182.5, df = 1,354, R2 = 0.34, P < 0.0001) (Fig. 7).
Number of insecticide sprays used to manage lepidopteran larvae across mesoregions and crop seasons in Brazil. Dashed line represents the start of commercial planting of Cry1Ac soybean in Brazil. Data on use of insecticide sprays to manage lepidopteran larvae across mesoregions for the 2013 to 2019 cropping seasons were obtained from the AMIS Kleffmann Group database (2013–2018) and BIP Spark (2019).
Discussion
This work presents the most extensive geographic assessment of lepidopteran pests on Brazil's soybean fields that covers more than 35 million hectares. C. includens was the main lepidopteran species occurring in non-Bt soybean (RR) fields in our study, being present in all the regions evaluated. This species was considered a secondary pest of soybean until the early 2000s. Its relevance in soybean likely increased because of changes in cultivation systems (i.e., no-till and cultivation of multiple crop and non-crop hosts of this species) and a decline in the adoption of IPM practices7,37. Both cotton and soybean have been documented as suitable hosts of C. includens38,39,40,41. The large increase in soybean cultivation area in Brazil seems to be a particularly important component in C. includens adaptation31. Soybean farms now are predominant in the agricultural landscape, narrowing the host plant availability to C. includens in some regions31. Increases in cultivation of other crop and non-crop hosts of C. includens also may have created a “green bridge” favoring the growth and spread of populations7,31. This likely increased the selective pressure of insecticides and Bt soybean plants, leading to higher resistance risk for these control tactics. C. includens prefers to feed on the lower and mid canopies of soybean plants, making it difficult to manage with insecticide sprays in the first place42. The resistance of C. includens populations to pyrethroids and chitin synthesis inhibitors has further contributed to the increased prevalence of this pest43,44. However, even under this high-resistance-risk scenario, our data showed that Cry1Ac soybean continues to be effective at controlling this pest. The near-high-dose level of the Cry1Ac Bt soybean MON 87701 × MON 89788 against C. includens12 and the low initial resistance allele frequency45 in C. includens have been key to managing the risk of Bt resistance in this pest.
A. gemmatalis was recognized as a major defoliating insect associated with soybean fields in Brazil, requiring an average of 2 insecticide applications every season46. Our results showed that the abundance of A. gemmatalis was lower than that of C. includens before and after the commercial launch of Cry1Ac soybean (2011–2014 and 2019–2020 cropping seasons). In the 2011 to 2014 sampling, A. gemmatalis was the second most abundant pest after C. includens, confirming that these two were the major pests of soybean in the early part of the decade. A. gemmatalis feeds primarily on leguminous plants (at least 34 species within Fabaceae family) and on only three other families (Begoniaceae, Poaceae and Malvaceae), with five species in these families serving as larval hosts47,48. This relatively narrow host range, in combination with the high efficacy of Cry1Ac soybean against A. gemmatalis12, may have contributed to reduced abundance of this species in Brazil. Our analysis showed that A. gemmatalis made a significant contribution to defoliation and is widely distributed in non-Bt soybean fields, so it is important to monitor for this species. In contrast, the low relative abundance of A. gemmatalis in Cry1Ac soybean showed that the pest is being effectively controlled by this technology in Brazil eight years after commercial launch.
Helicoverpa spp. was found at relatively low abundance. Within the Helicoverpa species found in Brazil, H. armigera are prevalent on dicotyledonous hosts such as soybean and cotton and H. zea on maize49,50,51,52. Therefore, most of the Helicoverpa spp. larvae in our collections are likely to be H. armigera. Although this species was first reported causing damage in soybean in Brazil53, the suitability of cotton as a host plant seems to be higher than soybean, as evidenced by higher larval viability and net reproductive rate52,54. The broad cultivation of row crops (e.g. soybean, cotton and maize) and availability of non-crop hosts across Brazil throughout the year, in combination with the high polyphagia of H. armigera, may be shaping the dynamics of this pest5,52. The relatively low abundance of this pest in soybean in Brazil also may be related to the broad adoption of Cry1Ac soybean, which is highly efficacious against this pest14,52.
The prevalence of S. frugiperda in non-Bt soybean was higher in 2019–2020 than in 2011–2014, increasing from 0.12% to more than 13%. S. frugiperda is a major pest of maize and cotton30,55,56,57, but recently has been also reported as a pest of soybean in Brazil6,58,59. The occurrence of S. frugiperda on soybean is favored by its ability to develop on several host plants60, high dispersal and migratory capacity61,62, high reproductive potential63, adaptation to Brazilian crop systems with availability of suitable hosts throughout the year30, and resistance to several classes of insecticides64,65,66,67,68,69. Although soybean plants produce proteinase inhibitors, S. frugiperda can adapt by altering the composition of proteolytic enzymes in the midgut70. S. frugiperda also expresses detoxification gene families that enable rapid response to plant secondary metabolites71. The abundance of green plant material provided by winter cover crops such as millet, which are highly suitable for S. frugiperda57, can also contribute to keeping populations of this pest at reasonably high levels throughout the year, creating a “green bridge” enabling dispersal and/or migration among hosts. Another factor that may be influencing S. frugiperda population growth and increasing its occurrence in soybean is the recent increase in winter maize area, where maize is rotated with soybean, and decrease in summer maize acreage in Brazil5. Currently, winter maize is planted on more than 13 million hectares, representing most of the maize planted in Brazil; in contrast, summer maize represented 4.3 million hectares in 20205. The removal of a significant number of maize plants from the landscape during the summer season may have triggered S. frugiperda to more frequently explore and colonize other suboptimal but readily available hosts such as soybean. Our analyses indicate that S. frugiperda could contribute to defoliation in soybean fields, though its capacity to defoliate soybean is lower than some other Spodoptera species.
S. eridania was more abundant than S. frugiperda during the late reproductive stages of soybean. S. eridania is also a polyphagous pest, reported to be capable of feeding on 202 host plant species72. Compared to cotton, soybean is a less suitable host for S. eridania, leading to lower pupal survivorship when consumed exclusively73. However, in the soybean–cotton farming system in the Cerrado region of Brazil (in the Central-West of the country), this pest may be of greater importance because it can find a continuous source of food in these two crops73. Sampling of lepidopteran larvae from soybeans at four locations in Mato Grosso do Sul State in 2011/12 showed that Spodoptera species represented about 10% of lepidopteran larvae in the samples74. Another study at one location in 2015/16 showed that Spodoptera accounted for 5% of the total lepidopteran larval sample: among these larvae, more than 50% were S. eridania75. In addition to feeding on leaves, Spodoptera species can feed on soybean pods6, which may have contributed to the higher density of S. eridania observed at the late reproductive stage in our collections.
S. cosmioides was at lower abundance than the other two Spodoptera species mentioned above, but its capacity to defoliate the soybean crop was greater than that of any other species in our collections. Its high capacity to cause damage has been demonstrated under laboratory conditions. For example, S. cosmioides was able to defoliate nearly twice the area defoliated by A. gemmatalis, S. eridania or S. frugiperda6. S. cosmioides is also a polyphagous pest capable of feeding on 126 plant species76. Soybean and cotton are conducive to development of this species, but maize does not allow its larval development77,78. Soybean and cotton also are preferred hosts for oviposition of the species when compared to oats, wheat and maize78. Therefore, the monitoring of this pest in soybean and cotton fields is important to prevent yield loss due to significant defoliation or pod damage.
The Spodoptera species are not controlled by Cry1Ac soybean, so their presence is expected in both Cry1Ac soybean and non-Bt soybean fields15. Larvae of Spodoptera species predominated in Cry1Ac soybean fields in our study, and the numbers of these three species were similar between Cry1Ac soybean and non-Bt soybean fields. Therefore, any differences in lepidopteran control tactics adopted by growers in Cry1Ac soybean and non-Bt soybean fields have not resulted in an increase in density of these Spodoptera species on Cry1Ac soybean relative to non-Bt soybean fields.
Abundance of both C. virescens and R. nu was low in our samples from the 2019 and 2020 seasons. Both species are considered pests of soybeans in Brazil33. Combined, these species represented less than 2.3% and 1.0% of the samples at the early and late reproductive stages in non-Bt soybean, respectively. Low abundance of these species was also observed in the 2011–2014 samples. C. virescens is a major pest in cotton and used to be observed attacking soybean in the central region of Brazil79. Cry1Ac soybean meets the high-dose concept for C. virescens13 and continues to provide effective control according to our results. In the USA, this pest is also considered an important pest of cotton, and the high adoption of Bt cotton in the USA may have reduced its abundance over large areas80. In Brazil, high adoption of both Bt cotton and Cry1Ac soybean could also be influencing the abundance of C. virescens. R. nu occurs predominantly in southern South America, and this pest is an important defoliator of soybean in Argentina81. R. nu is more adapted to subtropical and temperate regions than to tropical regions and is favored by higher latitudes and altitudes41. However, this pest has been reported from southern (Rio Grande do Sul and Paraná) to central regions of Brazil (Distrito Federal)75,82,83,84.
Despite fluctuations in lepidopteran pest abundance across regions and cropping seasons, C. includens and A. gemmatalis continue to be the main lepidopteran pests on non-Bt soybean in Brazil. The absence or very low density of these two species and reduced levels of defoliation on Bt soybean observed across regions and seasons indicate that Cry1Ac soybean still provides effective protection against these species. A significant reduction in the number of insecticide sprays to manage lepidopteran larvae has occurred over mesoregions and crop seasons, indicating that increasing adoption of Cry1Ac soybean has effective managed and apparently suppressed C. includens and A. gemmatalis populations across soybean-growing regions. Assessing the environmental impact of this reduction in lepidopteran sprays would be worthwhile. Suppression of target pests after a long period of use of Bt technologies has been documented in P. gossypiella, O. nubilalis and H. zea in the USA18,19,21 and H. armigera in China20. However, Spodoptera species are not controlled by Cry1Ac soybean15, and consequently they can be found on both Cry1Ac and non-Bt soybean. The occurrence of Spodoptera species, which were historically considered as secondary pests of soybeans in Brazil, on Cry1Ac and non-Bt soybeans may be associated with the high efficacy of the Cry1Ac soybean against target species (i.e., C. includens and A. gemmatalis) and the resulting reduction in insecticide use in soybeans fields in Brazil (Fig. 7). The challenge posed by secondary pests such as Spodoptera species highlights the need to develop Bt soybean technologies with novel modes of action59,85,86. When available, Bt soybean technologies with diverse modes of action will enhance pest management systems for soybean in Brazil.
It is also important to emphasize that the planting of refuge is crucial to the management of Bt crop pests. In Brazil, soybean, maize and cotton are planted simultaneously or in succession within the Cerrado landscape30. The proteins used in Bt soybean, maize and cotton overlap to a large degree and several of the most important target pests feed on two or all three of these crops e.g., S. frugiperda, H. armigera and C. includens, as discussed herein. Cross-crop resistance is a threat to their management. Indeed, resistance of S. frugiperda to Bt maize is already affecting the efficacy of Bt soybean and cotton due to cross resistance resulting from shared or similar Bt proteins among technologies59,87. Therefore, adherence to refuge recommendations for Bt maize, cotton and soybean is necessary to enhance durability of current and future Bt technologies in this multi-crop agroecosystem.
Overall, our study provides a large-scale assessment of Cry1Ac soybean field efficacy and demonstrates that its pest control benefits are being sustained. Cry1Ac soybean has provided Brazilian farmers with eight years of consistent protection against damage from the primary lepidopteran soybean pests (C. includens and A. gemmatalis). However, Cry1Ac soybean needs to be viewed as one tool within the pest management toolbox and should be integrated with other effective control tactics.
References
CONAB—Companhia Nacional de Abastecimento. Acompanhamento da Safra Brasileira de Grãos. V.7 - SAFRA 2019/20 - N. 12 - Décimo segundo levantamento. https://www.conab.gov.br/info-agro/safras (2020).
Panizzi, A. R. & Corrêa-Ferreira, B. S. Dynamics in the insect fauna adaption to soybeans in the tropics. Trends Entomol. 1, 71–88 (1997).
Cattelan, A. J. & Dall’Agnol, A. The rapid soybean growth in Brazil. Oilseeds Fats Crops Lipids 25, D102 (2018).
Freitas, P. L. & Landers, J. N. The transformation of agriculture in Brazil through development and adoption of Zero Tillage Conservation Agriculture. Int. Soil Wat. Cons. Res. 2, 35–46 (2014).
Brookes, G., Taheripour, F. & Tyner, W. E. The contribution of glyphosate to agriculture and potential impact of restrictions on use at the global level. GM Crops Food 8, 216–228 (2017).
Bueno, R. C. O. F., Bueno, A. F., Moscardi, F., Parra, J. R. P. & Hoffmann-Campo, C. B. Lepidopteran larva consumption of soybean foliage: Basis for developing multiple-species economic thresholds for pest management decisions. Pest Manag. Sci. 67, 170–174 (2011).
Panizzi, A. R. History and contemporary perspectives of the integrated pest management of soybean in Brazil. Neotrop. Entomol. 42, 119–127 (2013).
Bortolotto, A. et al. The use of soybean integrated pest management in Brazil: A review. Embrapa Soja-Artigo em periódico indexado (ALICE) Agron. Sci. Biotechnol. 1, 25–32 (2015).
CIB; AGROCONSULT. Impactos Econômicos e Sócio-ambientais da Tecnologia de Plantas Resistentes a Insetos no Brasil - Análise Histórica, Perspectivas e Desafios Futuros. http://apps.agr.br/wp-content/uploads/2018/12/Impactos-do-Milho-Bt-no-Brasil.pdf (2018).
Brookes, G. The farm level economic and environmental contribution of Intacta soybeans in South America: The first five years. GM Crops Food 9, 140–151 (2018).
Macrae, T. C. et al. Laboratory and field evaluations of transgenic soybean exhibiting high-dose expression of a synthetic Bacillus thuringiensis cry1A gene for control of Lepidoptera. J. Econ. Entomol. 98, 577–587 (2005).
Bernardi, O. et al. Assessment of the high-dose concept and level of control provided by MON 87701× MON 89788 soybean against Anticarsia gemmatalis and Pseudoplusia includens (Lepidoptera: Noctuidae) in Brazil. Pest Manag. Sci. 68, 1083–1091 (2012).
Bernardi, O. et al. High levels of biological activity of Cry1Ac protein expressed on MON 87701× MON 89788 soybean against Heliothis virescens (Lepidoptera: Noctuidae). Pest Manag. Sci. 70, 588–594 (2014).
Dourado, P. M. et al. High susceptibility to Cry1Ac and low resistance allele frequency reduce the risk of resistance of Helicoverpa armigera to Bt soybean in Brazil. PLoS ONE 11, e0161388 (2016).
Bernardi, O. et al. Low susceptibility of Spodoptera cosmioides, Spodoptera eridania and Spodoptera frugiperda (Lepidoptera: Noctuidae) to genetically-modified soybean expressing Cry1Ac protein. Crop Prot. 58, 33–40 (2014).
Edgerton, M. D. et al. Transgenic insect resistance traits increase corn yield and yield stability. Nat. Biotechnol. 30, 493–496 (2012).
Lu, Y., Wu, K., Jiang, Y., Guo, Y. & Desneux, N. Widespread adoption of Bt cotton and insecticide decrease promotes biocontrol services. Nature 487, 362–365 (2012).
Carrière, Y. et al. Long-term regional suppression of pink bollworm by Bacillus thuringiensis cotton. Proc. Natl. Acad. Sci. USA 100, 1519–1523 (2003).
Hutchison, W. D. et al. Areawide suppression of European corn borer with Bt maize reaps savings to non-Bt maize growers. Science 330, 222–225 (2010).
Wu, K. M., Lu, Y. H., Feng, H. Q., Jiang, Y. Y. & Zhao, J. Z. Suppression of cotton bollworm in multiple crops in China in areas with Bt toxin–containing cotton. Science 321, 1676–1678 (2008).
Dively, G. P. et al. Regional pest suppression associated with widespread Bt maize adoption benefits vegetable growers. Proc. Natl. Acad. Sci. USA 115, 3320–3325 (2018).
Lu, Y. et al. Mirid bug outbreaks in multiple crops correlated with wide-scale adoption of Bt cotton in China. Science 328, 1151–1154 (2010).
Zhao, J. H., Ho, P. & Azadi, H. Benefits of Bt cotton counterbalanced by secondary pests? Perceptions of ecological change in China. Environ. Monit. Assess. 173, 985–994 (2011).
Gould, F. Sustainability of transgenic insecticidal cultivars: Integrating pest genetics and ecology. Annu. Rev. Entomol. 43, 701–726 (1998).
Van Rensburg, J. B. J. First report of field resistance by the stem borer, Busseola fusca (Fuller) to Bt-transgenic maize. S. Afr. J. Plant Soil 24, 147–151 (2007).
Storer, N. P. et al. Discovery and characterization of field resistance to Bt maize: Spodoptera frugiperda (Lepidoptera: Noctuidae) in Puerto Rico. J. Econ. Entomol. 103, 1031–1038 (2010).
Dhurua, S. & Gujar, G. T. Field-evolved resistance to Bt toxin Cry1Ac in the pink bollworm, Pectinophora gossypiella (Saunders) (Lepidoptera: Gelechiidae), from India. Pest Manag. Sci. 67, 898–903 (2011).
Gassmann, A. J., Petzold-Maxwell, J. L., Keweshan, R. S. & Dunbar, M. W. Field-evolved resistance to Bt maize by western corn rootworm. PLoS ONE 6, e22629 (2011).
Farias, J. R. et al. Field-evolved resistance to Cry1F maize by Spodoptera frugiperda (Lepidoptera: Noctuidae) in Brazil. Crop Prot. 64, 150–158 (2014).
Fatoretto, J. C., Michel, A. P., Silva Filho, M. C. & Silva, N. Adaptive potential of fall armyworm (Lepidoptera: Noctuidae) limits Bt trait durability in Brazil. J. Integr. Pest Manag. 8, 17 (2017).
Silva, C. S. et al. Population expansion and genomic adaptation to agricultural environments of the soybean looper, Chrysodeixis includens. Evol. Appl. 13, 2071–2085 (2020).
Herzog, D. C. Sampling soybean looper on soybean. In Sampling Methods in Soybean Entomology (eds Koogan, M. & Herzog, D. C.) 141–168 (Springer, 1980).
Sosa-Gómez, D. R. et al. Manual de Identificação de Insetos e Outros Invertebrados da Cultura da Soja (Embrapa Soja-Documentos (INFOTECA-E), 2014).
Gilligan, T. M. & Passoa, S. C. LepIntercept–An identification resource for intercepted Lepidoptera larvae. (Identification Technology Program (ITP), 2014). http://idtools.org/id/leps/lepintercept/key.html.
R Core Team. R: A Language and Environment for Statistical Computing. (R Foundation for Statistical Computing, 2020). https://www.R-project.org/.
Kaster, M. & Farias, J. R. B. Regionalização dos Testes de Valor de Cultivo e Uso e da indicação de Cultivares de Soja-terceira Aproximação (Embrapa Soja-Documentos (INFOTECA-E), 2012).
Sosa-Gómez, D. R., Delpin, K. E., Moscardi, F. & Nozaki, M. D. H. The impact of fungicides on Nomuraea rileyi (Farlow) Samson epizootics and on populations of Anticarsia gemmatalis Hübner (Lepidoptera: Noctuidae), on soybean. Neotrop. Entomol. 32, 287–291 (2003).
Specht, A., Paula-Moraes, S. V. & Sosa-Gómez, D. R. Host plants of Chrysodeixis includens (Walker) (Lepidoptera, Noctuidae, Plusiinae). Rev. Bras. Entomol. 59, 343–345 (2015).
Andrade, K. et al. Bioecological characteristics of Chrysodeixis includens (Lepidoptera: Noctuidae) fed on different hosts. Austral. Entomol. 55, 449–454 (2016).
Moonga, M. N. & Davis, J. A. Partial life history of Chrysodeixis includens (Lepidoptera: Noctuidae) on summer hosts. J. Econ. Entomol. 109, 1713–1719 (2016).
Specht, A. et al. Biotic potential and life tables of Chrysodeixis includens (Lepidoptera: Noctuidae), Rachiplusia nu, and Trichoplusia ni on soybean and forage turnip. J. Insect Sci. 19, 8 (2019).
Zulin, D., Ávila, C. J. & Schlick-Souza, E. C. Population fluctuation and vertical distribution of the soybean looper (Chrysodeixis includes) in soybean culture. Am. J. Plant Sci. 9, 1544–1556 (2018).
Stacke, R. F. et al. Field-evolved resistance to chitin synthesis inhibitor insecticides by soybean looper, Chrysodeixis includens (Lepidoptera: Noctuidae), in Brazil. Chemosphere 259, 127499 (2020).
Stacke, R. F. et al. Inheritance of lambda-cyhalothrin resistance, fitness costs and cross-resistance to other pyrethroids in soybean looper, Chrysodeixis includens (Lepidoptera: Noctuidae). Crop Prot. 131, 105096 (2020).
Yano, S. A. et al. High susceptibility and low resistance allele frequency of Chrysodeixis includens (Lepidoptera: Noctuidae) field populations to Cry1Ac in Brazil. Pest Manag. Sci. 72, 1578–1584 (2016).
Silva, M. T. B. & Moscardi, F. Field efficacy of the nucleopolyhedrovirus of Anticarsia gemmatalis Hübner (Lepidoptera: Noctuidae): Effect of formulations, water pH, volume and time of application, and type of spray nozzle. Neotrop. Entomol. 31, 75–83 (2002).
Herzog, D. C. & Todd, J. W. Sampling velvetbean caterpillar on soybean. In Sampling Methods in Soybean Entomology (eds Koogan, M. & Herzog, D. C.) 107–140 (Springer, 1980).
Panizzi, A. R., Oliveira, L. J. & Silva, J. J. Survivorship, larval development and pupal weight of Anticarsia gemmatalis (Hübner) (Lepidoptera: Noctuidae) feeding on potential leguminous host plants. Neotrop. Entomol. 33, 563–567 (2004).
Leite, N. A., Alves-Pereira, A., Corrêa, A. S., Zucchi, M. I. & Omoto, C. Demographics and genetic variability of the new world bollworm (Helicoverpa zea) and the old world bollworm (Helicoverpa armigera) in Brazil. PLoS ONE 9, e113286 (2014).
Leite, N. A. et al. Pan-American similarities in genetic structures of Helicoverpa armigera and Helicoverpa zea (Lepidoptera: Noctuidae) with implications for hybridization. Environ. Entomol. 46, 1024–1034 (2017).
Sosa-Gómez, D. R. et al. Timeline and geographical distribution of Helicoverpa armigera (Hübner) (Lepidoptera, Noctuidae: Heliothinae) in Brazil. Rev. Bras. Entomol. 60, 101–104 (2016).
Dourado, P. M. et al. Host plant use of Helicoverpa spp. (Lepidoptera: Noctuidae) in the Brazilian agricultural landscape. Pest Manag. Sci. 77, 780–794 (2021).
Czepak, C., Albernaz, K. C., Vivan, L. M., Guimarães, H. O. & Carvalhais, T. First reported occurrence of Helicoverpa armigera (Hübner) (Lepidoptera: Noctuidae) in Brazil. Pesqui. Agropecu. Trop. 43, 110–113 (2013).
Gomes, E. S., Santos, V. & Ávila, C. J. Biology and fertility life table of Helicoverpa armigera (Lepidoptera: Noctuidae) in different hosts. Entomol. Sci. 20, 419–426 (2017).
Luttrell, R. G. & Mink, J. S. Damage to cotton fruiting structures by the fall armyworm, Spodoptera frugiperda (Lepidoptera: Noctuidae). J. Cotton Sci. 3, 35–44 (1999).
Martinelli, S., Barata, R. M., Zucchi, M. I., DeCastroSilva-Filho, M. & Omoto, C. Molecular variability of Spodoptera frugiperda (Lepidoptera: Noctuidae) populations associated to maize and cotton crops in Brazil. J. Econ. Entomol. 99, 519–526 (2006).
Barros, E. M., Torres, J. B., Ruberson, J. R. & Oliveira, M. D. Development of Spodoptera frugiperda on different hosts and damage to reproductive structures in cotton. Ent. Exp. Appl. 137, 237–245 (2010).
Silva, D. M. D. et al. Biology and nutrition of Spodoptera frugiperda (Lepidoptera: Noctuidae) fed on different food sources. Sci. Agric. 74, 18–31 (2017).
Machado, E. P. et al. Cross-crop resistance of Spodoptera frugiperda selected on Bt maize to genetically-modified soybean expressing Cry1Ac and Cry1F proteins in Brazil. Sci. Rep. 10, 1–9 (2020).
Montezano, D. G. et al. Host plants of Spodoptera frugiperda (Lepidoptera: Noctuidae) in the Americas. Afr. Entomol. 26, 286–300 (2018).
Nagoshi, R. N. & Meagher, R. L. Review of fall armyworm (Lepidoptera: Noctuidae) genetic complexity and migration. Fla. Entomol. 91, 546–554 (2008).
Westbrook, J. K., Nagoshi, R. N., Meagher, R. L., Fleischer, S. J. & Jairam, S. Modeling seasonal migration of fall armyworm moths. Int. J. Biometeorol. 60, 255–267 (2016).
Garcia, A. G., Ferreira, C. P., Godoy, W. A. & Meagher, R. L. A computational model to predict the population dynamics of Spodoptera frugiperda. J. Pest Sci. 92, 429–441 (2019).
Diez-Rodríguez, G. I. & Omoto, C. Herança da resistência de Spodoptera frugiperda (J.E. Smith) (Lepidoptera: Noctuidae) a lambda-cialotrina. Neotrop. Entomol. 30, 311–316 (2001).
Carvalho, R. A., Omoto, C., Field, L. M., Williamson, M. S. & Bass, C. Investigating the molecular mechanisms of organophosphate and pyrethroid resistance in the fall armyworm Spodoptera frugiperda. PLoS ONE 8, e62268 (2013).
Nascimento, A. R. B. et al. Genetic basis of Spodoptera frugiperda (Lepidoptera: Noctuidae) resistance to the chitin synthesis inhibitor lufenuron. Pest Manag. Sci. 72, 810–815 (2016).
Okuma, D. M. et al. Inheritance and fitness costs of Spodoptera frugiperda (Lepidoptera: Noctuidae) resistance to Spinosad in Brazil. Pest Manag. Sci. 74, 1441–1448 (2018).
Bolzan, A. et al. Selection and characterization of the inheritance of resistance of Spodoptera frugiperda (Lepidoptera: Noctuidae) to chlorantraniliprole and cross-resistance to other diamide insecticides. Pest Manag. Sci. 75, 2682–2689 (2019).
Lira, E. C. et al. Resistance of Spodoptera frugiperda (Lepidoptera: Noctuidae) to spinetoram: Inheritance and cross-resistance to spinosad. Pest Manag. Sci. 76, 2674–2680 (2020).
Paulillo, L. C. M. et al. Changes in midgut endopeptidase activity of Spodoptera frugiperda (Lepidoptera: Noctuidae) are responsible for adaptation to soybean proteinase inhibitors. J. Econ. Entomol. 93, 892–896 (2000).
Silva-Brandão, K. L. et al. Transcript expression plasticity as a response to alternative larval host plants in the speciation process of corn and rice strains of Spodoptera frugiperda. BMC Genom. 18, 1–15 (2017).
Montezano, D. G., Specht, A., Sosa-Gomez, D. R., Roque-Specht, V. F. & Barros, N. M. Immature stages of Spodoptera eridania (Lepidoptera: Noctuidae): Developmental parameters and host plants. J. Insect Sci. 14, 238 (2014).
Santos, K. B., Meneguim, A. M. & Neves, P. M. O. J. Biologia de Spodoptera eridania (Cramer) (Lepidoptera: Noctuidae) em diferentes hospedeiros. Neotrop. Entomol. 34, 903–910 (2005).
Justiniano, W., Fernandes, M. G. & Viana, C. L. T. P. Diversity, composition and population dynamics of arthropods in the genetically modified soybeans Roundup Ready® RR1 (GT 40-3-2) and Intacta RR2 PRO (MON87701 x MON89788). J. Agric. Sci. 6, 33 (2014).
Specht, A. et al. Owlet moths (Lepidoptera: Noctuoidea) associated with Bt and non-Bt soybean in the Brazilian savanna. Braz. J. Biol. 79, 248–256 (2019).
Specht, A. & Roque-Specht, V. F. Immature stages of Spodoptera cosmioides (Lepidoptera: Noctuidae): Developmental parameters and host plants. Zoologia 33, e20160053 (2016).
Habib, M. E. M., Paleari, M. L. & Amaral, M. E. C. Effect of three larval diets on the development of the armyworm, Spodoptera latifascia Walker, 1856 (Lepidoptera: Noctuidae). Rev. Bras. Zool. 1, 177–182 (1983).
Silva, D. M. et al. Biology of Spodoptera eridania and Spodoptera cosmioides (Lepidoptera: Noctuidae) on different host plants. Fla. Entomol. 100, 752–760 (2017).
Tomquelski, G. V. & Maruyama, L. C. T. Lagarta-da-macã em soja. Rev. Cultiv. 117, 20–22 (2009).
Blanco, C. A. Heliothis virescens and Bt cotton in the United States. GM Crops Food 3, 201–212 (2012).
Barrionuevo, M. J., Murúa, M. G., Goane, L., Meagher, R. & Navarro, F. Life table studies of Rachiplusia nu (Guenée) and Chrysodeixis (= Pseudoplusia) includens (Walker) (Lepidoptera: Noctuidae) on artificial diet. Fla. Entomol. 95, 944–951 (2012).
Specht, A. et al. Ocorrência de Rachiplusia nu (Guenée) (Lepidoptera: Noctuidae) em Fumo (Nicotiana tabacum L.) no Rio Grande do Sul. Neotrop. Entomol. 35, 705–706 (2006).
Trentin, L. B. et al. The complete genome of Rachiplusia nu nucleopolyhedrovirus (RanuNPV) and the identification of a baculoviral CPD-photolyase homolog. Virology 534, 64–71 (2019).
Perini, C. R. et al. Genetic structure of two Plusiinae species suggests recent expansion of Chrysodeixis includens in the American continent. Agric. For. Entomol. 23, 2502–3260 (2020).
Bacalhau, F. B. et al. Performance of genetically modified soybean expressing the Cry1A. 105, Cry2Ab2, and Cry1Ac proteins against key Lepidopteran pests in Brazil. J. Econ. Entomol. 113, 2883–2889 (2020).
Machado, E. P. et al. Survival and development of Spodoptera eridania, Spodoptera cosmioides and Spodoptera albula (Lepidoptera: Noctuidae) on genetically-modified soybean expressing Cry1Ac and Cry1F proteins. Pest Manag. Sci. 76, 4029–4035 (2020).
Horikoshi, R. J. et al. Effective dominance of resistance of Spodoptera frugiperda to Bt maize and cotton varieties: Implications for resistance management. Sci. Rep. 6, 1–8 (2016).
Acknowledgements
We thank the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES) (Finance Code 001) and scholarships provided by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) to ASC (311465/2018-8) and CO (#312086/2013-0).
Author information
Authors and Affiliations
Contributions
R.J.H., P.M.D., G.U.B., C.O., S.M., G.P.H. and A.S.C. conceived and designed the experiments; R.J.H., P.M.D., D.S.F., A.S.C. collected data; R.J.H., P.M.D., A.W., S.M., G.P.H. and A.S.C. performed the statistical analysis; R.J.H., P.M.D., G.U.B., C.O., D.S.F., A.W., S.M., G.P.H. and A.S.C. interpreted the results, discussed and wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Horikoshi, R.J., Dourado, P.M., Berger, G.U. et al. Large-scale assessment of lepidopteran soybean pests and efficacy of Cry1Ac soybean in Brazil. Sci Rep 11, 15956 (2021). https://doi.org/10.1038/s41598-021-95483-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-95483-9
This article is cited by
-
Identification of wormholes in soybean leaves based on multi-feature structure and attention mechanism
Journal of Plant Diseases and Protection (2023)
-
Refuge areas favor the presence of predators and herbivores in Bt soybean: a landscape perspective
Journal of Pest Science (2023)
-
Functional characterization of the pUceS8.3 promoter and its potential use for ectopic gene overexpression
Planta (2022)
-
Resistance status of lepidopteran soybean pests following large-scale use of MON 87701 × MON 89788 soybean in Brazil
Scientific Reports (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.